Sommaire
- Validation Strategy of Viral Decontamination Methods, a quick overview
- Antibody-Drug conjugate Manufacturing Techniques
- Robust and Convenient Single-use Processing
- Cahier Pratique – Quality by design applied to viral safety of Biologicals: Case studies & workshop discussion summary
- Mass spectrometry as a powerful tool for the characterisation of monoclonal antibodies in the context of comparability studies
- Chromatographie Continue : Solution d’amélioration des performances de procédés et « debottlenecking » des capacités de Bioproduction
- Protein A Affinity Chromatography for Efficient Fab Purification
- Enabling Higher Post Protein A Product Purity Using Novel Chromatographic Clarification Approach
Monoclonal Antibodies (mAbs) with selectivity towards antigens located on the surface of cancer cells have been successfully developed. Nonetheless, mAbs alone are generally not potent to kill the cancer cells and are therefore used in combination with classical chemotherapies.
The small molecule cytotoxic drugs used in chemotherapies effectively kill fast dividing cells. These include but are not restricted to cancer cells, meaning that side-effects are generally associated with conventional chemotherapies. Moreover, the most potent and potentially most effective drugs cannot be used because side-effects would be too severe. An antibody-Drug Conjugate (ADC) is the unique combination of a targeting monoclonal antibody, a stable linker, and a potent cytotoxic agent (1). It is designed to deliver anti-cancer agents directly to the tumor cell in a targeted manner to limit systemic exposure (Fig. 1).
Efforts in this field have led to the approval of two ADCs as drugs: Adcetris for the treatment of refractory Hodgkin lymphoma and anaplastic large-cell lymphoma and Kadcyla for the treatment of metastatic breast cancer. Auristatin and maytansine derivatives are used as cytotoxic agents, respectively. On average, 2 to 4 toxins are attached or conjugated to the mAb. In addition to the two commercial ADCs, close to forty are under investigation in many different cancer types.
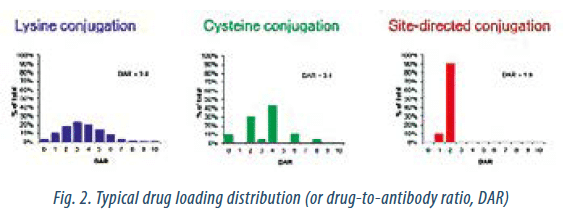
Conventional ADCs, where conjugation takes place either on lysine side-chains or to reduced cysteines, are heterogeneous. The average drug loading as well as the subpopulation must, however, be consistent from batch to batch. The conjugation process defines the product and the average loading must be the same for each batch. In recent years, site-selective conjugation technologies have been developed. Site-selective conjugations through mAb engineering, enzymatic conjugation reactions, or more selective chemical reactions is expected to afford more homogeneous ADCs with improved therapeutic properties (Fig. 2). The current clinical pipeline is, however, still dominated by random lysine and/or cysteine conjugation, which makes analytical characterization challenging.
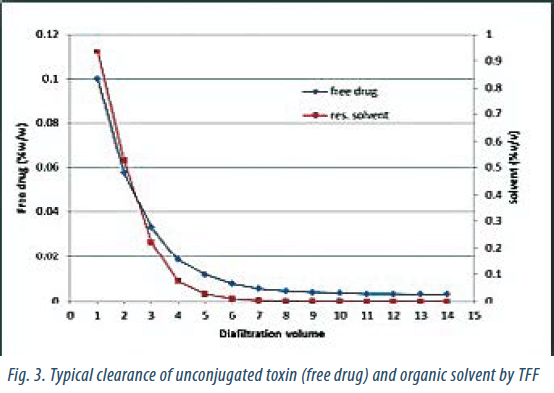
The conjugation reaction is followed by protein purification techniques to remove process-related contaminants (unconjugated toxin and residual solvent). During drug discovery, purification can be achieved through dialysis or with single-use Sephadex columns. These techniques are replaced by Tangential Flow Filtration (TFF) during process development and scale-up. Suitable conditions are developed, with the goal to achieve good impurity clearance, short TFF time and low aggregation rate. Once suitable membranes have been selected and the TFF parameters developed (feed flow, TMP, conc.), one can investigate how many diafiltration volumes are needed to clear out process-related impurities (Fig. 3). A well-developed TFF process is, in most cases, sufficient to reduce the amount of unconjugated toxin to an acceptable level. TFF, however, does not remove aggregates. If aggregate formation cannot be prevented during the conjugation process, a low-pressure chromatography purification step will be needed. In such cases, mixed-mode (CHT or MEP HyperCel) or ion-exchange resins are recommended.
Lonza has established dedicated teams and facilities to facilitate the scale-up of ADC projects. For clinical phase 1 and 2 material, our small scale assets are generally well suited both in terms of flexibility and vessel size (10 to 60 L). For phase 3 and beyond, the process is transferred into our commercial bioconjugation plant where we have 100 to 600 L stirred tanks (Fig. 4). These volumes are small compared to antibody manufacturing, explained by the higher process concentration, shorter batch time and high potency of the API.
Some safety challenges are associated with ADC manufacturing since one has to manufacture a highly toxic compound in a biopharmaceutical environment. A balance between occupational safety and cGMP requirements must be achieved. The typical ADC payloads are among the most toxic compounds ever produced and strict containment measures are needed. Typical occupational exposure levels for ADCs and their toxins are between 100 down to 1 ng/m3. But needless to say that when considering the potential benefits for cancer patients, it is gratifying to tackle the challenges associated with the ADC design, scale-up and manufacturing.
| Résumé Les conjugués anticorps-médicament, ou immunoconjugués cytotoxiques, représentent une nouvelle forme de thérapie anticancéreuse ciblée. Ils peuvent se lier à certains types de cellules cancéreuses dans lesquelles ils libèrent directement leurs agents chimiothérapeutiques. Différents types de conjugaison ont été développés, pouvant donner une nouvelle option de traitement pour les patients atteints de cancer. Lonza résume certains aspects liés à la production et à la purification de ces composés parmi les plus difficiles à manipuler. |

Laurent DUCRY – LONZA
laurent.ducry@lonza.com
Partager l’article
Bibliographie
(1) L. Ducry, Antibody-Drug Conjugates: Methods and Protocols, Springer, Humana Press, 2013