Sommaire
- ASTM E2500 : let's cross the Bridge !
- De l'étude du contexte à l'emploi de technologies de traitement des eaux adaptées
- Une nouvelle stratégie de gestion de l'eau en industrie pharmaceutique
- Cahier Pratique - Les boucles d'eau purifiée et la qualification. Focus sur la QP
- La mesure en continu dans l'eau à usage pharmaceutique (EUP)
- Candida auris, une espèce pathogène émergente "tueuse"
- Cleaning and disinfection program part of the lifecycle approach: a risk based rather arbitrarily imposed approach
- Eaux pharmaceutiques : les dessous de la maintenance des installations
- Pharma & the New Healthcare Economy. Ready or not ?
Cleaning and microbial contamination control are critical focus areas in the (bio) pharmaceutical and medical device industries. Robust cleaning and disinfection programs are required to prevent adulteration, cross-contamination, and microbial contamination of products. Consequently, the selection and number of disinfecting agents used, the frequency of application, and the rotation of (one or more) disinfectants along with a sporicidal agent should be scientifically justified by a formal microbial risk analysis (MRA). The MRA and the chosen cleaning and disinfection program should be supported by periodic review of relevant environmental monitoring (EM) data, and regular auditing of the cleaning and disinfection programs.
Inadequate cleaning and disinfection programs cause significant risk to patient safety, financial loss to the company and product recalls(1-5). Recurring microbial contamination generally results from inadequate cleaning and disinfection procedures associated with an ineffective root-cause investigation. Control of microbiological contamination and root- cause investigation (see CFR211.113 a and b, and CFR211.192) are amongst the top 10 most observed deficiencies by the FDA since 2012(6). The situation in Europe is not any different based on recent UK MHRA (Medicines Healthcare products Regulation Agency) deficiencies observed, and the noncompliance reports handed out by European Inspectors(7-9).
The objective of the article is first to present the different regulatory requirements and guidance regarding disinfectant rotation. Secondly, the article seeks to answer the question “Is the rotation of more than one disinfectant along with a sporicidal agent mandatory for cGMP compliance?”. To be able to fully answer the question, the process of establishing the number of disinfectants (one or more) will be discussed. Finally, the article reaffirms the importance of periodically reviewing microbial data and auditing practices to confirm the effectiveness of the cleaning and disinfection program including the disinfectant rotation and frequency choice.
1. Regulatory Requirements and Guidance on Disinfectant Rotation
The regulatory recommendations regarding rotation of disinfectants are the following:
1.1 Japan (JP) Guidance on the Manufacture of Sterile Pharmaceutical Products by Aseptic Processing (2006):
- “The program should contain procedures for screening and classifying bacterial isolates in each manufacturing ”
- “Cleaning agents and disinfectants should be validated for their appropriateness and reliability in removing contaminants prior to use. Cleaning and disinfection efficacy should be assessed and confirmed based on type and count of microorganisms characterized by periodic environmental monitoring”
- “If selected disinfectants might have inferior efficacy against microorganisms isolated from the environment, the efficacy should be reevaluated and the replacement with or alternate use of different disinfectants should be considered and implemented, as appropriate”
- “If environmental monitoring data indicate or suggest the presence of spore-forming bacteria or fungi, suitable sporicides or fungicides should be selected for disinfection.”
1.2 United States Food and Drug Administration (US FDA) guidance on aseptic manufacturing (2004):
- “… Routinely used disinfectants should be effective against the normal microbial vegetative flora recovered from the facility…”
- “…Therefore, a sound disinfectant program also includes a sporicidal agent, used according to a written schedule and when environmental data suggest the presence of sporeforming organisms.”
- “…If indicated, microorganisms associated with adverse trends can be investigated as to their sensitivity to the disinfectants employed in the cleanroom in which the organisms were isolated.”
1.3 Brazil National Health Surveillance Agency (ANVISA) resolution – RDC n.17 (2010):
- “Article 315. The sanitation of clean areas is particularly important in the manufacture of sterile products.
§1 These areas should be cleaned and sanitized frequently, according to a specific program approved by Quality Assurance.
§2 The areas should be monitored regularly to detect the emergence of resistant microorganisms.”
1.4 European Medicines Agency (EMA) EudraLex Annex 1 (2008):
- “61. The sanitation of clean areas is particularly important. They should be cleaned thoroughly in accordance with a written programme. Where disinfectants are used, more than one type should be employed. Monitoring should be undertaken regularly in order to detect the development of resistant strains”. The Pharmaceutical Inspection Convention Pharmaceutical Inspection Co-Operation (PIC/S) – PE 009-13 Annexes (2017) requirements regarding disinfectant rotation are similar to the EudraLex Annex 1 (2008).
Note that based on these excerpts, the reader might be confused about the rotation definition. Is it one disinfectant alternated with the use of a second disinfectant? Or, is it one or more than one disinfectant with a sporicidal agent? Several articles defining rotation have already been published(10-13). Different nongovernmental organizations have published recommendations on cleaning and disinfection programs design and disinfectant rotation(14-16). The rotation definition should be adequately defined in the cleaning and disinfection procedures.
General chapter from the pharmacopeias or technical documents from nongovernmental organization are not mandatory requirements. However, this information might include some recommendations that should help firms to meet cGMPs:
1. United States Pharmacopeia (USP) <1072>:
- “The rotation of an effective disinfectant with a sporicide is encouraged. It is prudent to augment the daily use of a bactericidal disinfectant with weekly (or monthly) use of a sporicidal agent.”
- “Other disinfection rotation schemes may be supported on the basis of a review of the historical environmental monitoring data. ”
- “…the most frequently isolated microorganisms from an environmental monitoring program may be periodically subjected to use-dilution testing with the agents used in the disinfection program to confirm their susceptibility, as there are real differences among different species in resistance to the lethal effects of different sanitizers.”
2. Parenteral Drug Association (PDA) Technical Report (TR) 70:
- “Today, most firms use a system whereby a disinfectant is rotated with a sporicide to more effectively reduce the bioburden levels.”
- “All rotation systems should be evaluated via the use of area classification, environmental monitoring data, and/or risk assessment.”
The regulatory guidelines agree on the idea that a routine disinfectant must be active against vegetative microbial flora recovered from the facility or isolates. The effectiveness of the disinfectants and the sporicides must be demonstrated through a disinfectant validation (laboratory testing such as suspension and coupon testing and in-situ testing) and confirmed through environmental monitoring and data trending over time. Finally, the EM program should be designed to detect most frequent isolate microorganisms and worst- case microorganisms (e.g. spores or fungi) or “resistant” strains/microorganisms. The terminology used by the USP “most frequent isolate” is considered as more accurate for (bio)pharmaceutical and medical device firms than the term “resistant microorganism.” It is expected that where EM data shows the presence of spore-forming organisms, that sporicidal agent will be used. Note that this last statement is not specified in the EU GMP Annex 1 revised in 2008, but the author believes it should be stated in the future Annex 1 revision to align with the other regulatory recommendations.
Finally, the regulatory guidelines are not alignedregardingthenumberofdisinfectants required in conjunction with a sporicidal agent (disinfectant rotation). However, the USA FDA, JP and ANVISA guidelines expect the manufacturer to justify the number of disinfectants and the rotation based on EM data review. However, the EU GMP requires the use of at least more than one type of disinfectant, where a disinfectant is used(15, 22). In addition, the draft Annex 1 available for public consultation propose that when “More than one type of disinfecting agent should be employed, and should include the periodic use of a sporicidal agent.”. Isn’t this paradoxical with the quality risk management principle promoted in the different recent EU guidelines revisions? The cleaning and disinfection program should be considered as part of the EM program lifecycle approach. Consequently, the number of disinfectants used along with a sporicidal agent should be justified by a formal MRA, periodic EM data review and periodic auditing of the cleaning and disinfection procedures rather than being arbitrarily imposed.
2. Lifecycle Approach to Confirm the Disinfectant Rotation Programs
Disinfectant use is unlikely to lead to microbial resistance development(13). Sutton concludes that “the probable scenario for selection of a development of resistant variant would require exposure of an extremely large number of cells (in excess of 1,000,000 CFU) to a low level of the toxic chemical.”(13). Such circumstances should not arise in a typical clean-room. Also, Sutton states that “selection of mutants that are resistant to in-use levels of disinfectants has not been shown to happen in cleanroom settings. Literature reports of resistance to in-use levels are restricted to descriptions of survival of specific microorganisms in contaminated solutions”.
Therefore, the dilution, the filtration, and the storage must be performed effectively to avoid microbial contamination of the disinfectant solution. Finally, the open container stability (use shelf life), the bioburden or the sterility (if used in grade A/B or ISO5) should be monitored during a predefined period of use to confirm the absence of microbial contamination(15). To date, there have been no conclusive data showing resistance by microorganisms to these chemicals(14).
The idea behind disinfectant rotation is to cover the largest microbial spectrum. The disinfectant rotation frequency should be based on the historical EM data trending over time and the disinfectants’ efficacy profile. Based on this, the sole routine use of a sporicide with proven microbial efficacy against facility flora can be implemented without rotation(14,16). However, since a sporicidal agent has an inherent corrosive nature, it is typically not recommended for daily use(17). Therefore, if one broad-spectrum disinfectant along with a periodic use of a sporicidal agent covers the largest microbial spectrum and is effective to control microbial contamination, adding another broad- spectrum disinfectant may not add value. The rotation of two disinfectants with the same microbial efficacy profile is baseless practice. Rotation of two disinfectants should only occur with two different types of chemistries, or similar chemistries with two distinct microbial efficacy profiles, due to other aspects of the formulations. The complementary microbial actions should ensure that the alert level is not reached.
An effective disinfectant rotation program should take into consideration the following steps(14, 17, 21):
- Routine disinfectants should be used daily for cleaning and disinfection of non- product contact equipment to effectively kill vegetative microorganisms and remove soils, while minimizing the risk to personnel and cleanroom surfaces. The use of a detergent (for the cleaning step) should be defined based on the soil level of clean-rooms surfaces, the composition and effectiveness of the disinfectants, e.g., a surfactant in the composition will help to clean and disinfect.
- A sporicidal agent should be used periodicallytoachievekillofsporeforming bacteria (e.g., Bacillus species). The frequency of use should be based on the MRA results, area classifications and historical EM data. Note that the frequency of use of a sporicidal agent might be higher inside a restricted access barrier or isolator (ISO 5 or grade A area) than in a lowest grade classification. This situation is justified based on the area classification, microbial specification and the cleanness of the non-product contact surfaces inside an ISO 5 or grade A/B As a consequence, it justifies a reduction in the use of routine disinfectants.
- Disinfectant and sporicidal agent residue should be periodically rinsed using 70% Isopropanol (IPA) or Water For Injection (WFI). The rinse frequency should be set based on a visual inspection and tactile observations of the surfaces in the cleanroom. There are certainly some cases where residues become such an issue that a cleaner is needed (e.g., sticky, tacky, or slippery floors or doors) and then the cleaner residue is rinsed using purified water or WFI. Note that the periodic rinse could be integrated into the cleaning step when a detergent is used prior disinfection.
The number of disinfectants, their rotation, and program frequency should be justified based on the microbial risk assessment results, disinfectant and sporicide microbial efficacy profiles, area classification, and review of the historical EM data. However, for a new or entirely revamped facility, the justification should be based on the MRA results, disinfectants and sporicides microbial efficacy profiles and the classification area.
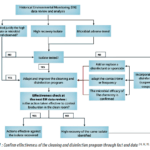
The environmental control program, including EM program, cleaning and disinfection program, qualification and periodic re-testing of the disinfectants should be looked at as a lifecycle approach (feedback loop). As a matter of fact, the historical EM data review, Figure 1, is one of the triggers to adapt or improve the cleaning and disinfection program and confirm that the disinfectant and sporicide microbial efficacy profile and spectrum is adequate.
Finally, periodic historical EM data review and analysis must be performed to:
- Confirm the absence of the increase in excursions from the previous historical EM data analysis.
- Identify the specific re-occurrence of trend or adverse events (e.g., the recurring deviation with the same microorganism or recurring excursion on the same sampling position).
- Identify specific worst-case microorganism.
- Categorize the source of isolate and contamination factors.
- Confirm the absence of a high occurrence of microorganisms of concern or microorganisms considered as objectionable or those under official scrutiny, e.g., Burkholderia cepacia(18).
Based on Figure 1 (page 41), conclusion and actions cannot be drawn based only on historical EM data review and analysis. The conclusion and actions could only be confirmed if supported by an audit of the cleaning and disinfection procedure. The goal of the audit is to demonstrate that the personnel and the microbial contamination control practices are compliant with the defined procedures. Moreover, the audit aims to confirm the absence of improper practices in the manufacturing area, e.g., ineffective chemistry is applied (e.g., use of alcohol against spore-forming microorganism), suboptimal concentrations, errors in dilution, wet contact times not in line with the disinfectant qualification data report or presence of visible residue (e.g., dust) affecting the efficacy of the disinfectant.
Finally, based on the historical EM data analysis and the audit results, the team can confirm if the cleaning and disinfection program should be adapted or improved, if a disinfectant or sporicide should be replaced or added or if the contact time or frequency should be reviewed. The approach mentioned above corresponds more to a life-cycle risk-based approach rather than arbitrary approach.
Conclusion
Depending on the regulatory guidelines, it is expected that at least one disinfectant be used to control routine vegetative microbial flora. Also, a periodic useofasporicidalagentisjustified if the historical environmental monitoring data confirms the presence of spore former microorganisms.
Today, most (bio)pharmaceutical firms use a program where a disinfectantisusedroutinely,such as daily, and is complemented by the use of a sporicide to more efficiently reduce the bioburden levels(14). Finally, the use of a disinfectant with a sporicide is considered superior or is encouraged over the rotation of multiple disinfectants(14, 16).
Effective risk management principles must be integrated into the product lifecycle to minimize microbial contamination and ensure the safety, quality, and efficacy of the product. Consequently, the manufacturer should incorporate the environmental monitoring program including the cleaning and disinfection program into the risk management strategy used and the product lifecycle to confirm product and the patient safety. Therefore, the goal of cleaning and disinfection program should be beyond elementary regulatory compliance.
The disinfectant rotation and rotation frequency must be based on the results of the microbial risk assessment, the chemical agent microbial efficacy profile, periodic audit and the historical environmental monitoring data.
Finally, compliance will be reached if the cleaning and disinfection program is capable of controlling the bioburden level in the clean rooms to acceptable levels.

Walid EL AZAB – STERIS
Responsable du service technique de la division life science de Steris, Walid a occupé divers postes, y compris la gestion d’équipe, dont celui de responsable projet, responsable de préparation et de gestion d’audit réglementaire, responsable qualité et réglementaire, et Personne Qualifiée (QP). Walid est titulaire d’un master en sciences pharmaceutiques industrielles de l’Université de Liège et est certifié ceinture verte (green belt) en amélioration continue.
walid_elazab@steris.com
Partager l’article
References
[1] Macher, Business Case for Quality. Pharmaceutical Quality Systems (ICH Q10) Conference. (2011)
[2] Sutton, S and L , A Review of Reported Recalls Involving Microbiological Control 2004-2011 with Emphasis on FDA Considerations of “Objectionable Organisms”, American Pharmaceutical Review, 15, 42-57, (2012).
[3] Hayes, R.J. Cost of Quality (CoQ) – An Analysis of the Cost of Maintaining a State of (2014) Int. Pharm. Ind. 6(1), 74-76.
[4] Cundell, Mold Monitoring and Control in Pharmaceutical Manufacturing Areas. (2016) Am. Pharm. Rev., 19(5).
[5] $18.2 million in settlement over mold at manufacturing facility, access on Jul 1, 2017 at: http://www.modernhealthcare. com/article/20170112/NEWS/170119945
[6] United States Food and Drug Administration (US FDA), Summary of Inspectional Observations by Fiscal Year, access on May 25, 2017 at: https://www.fda.gov/ICECI/Inspections.htm
[7] Medicines & Healthcare products Regulatory, MHRA GMP Inspection Deficiency Data Trend 2015, access on May 25, 2017 at: https://www.gov.uk/government/uploads/system/ uploads/attachment_data/file/582841/MHRA_GMP. pdf
[8] Medicines & Healthcare products Regulatory, MHRA GMP Inspection Deficiency Data Trend 2016, access on May 25, 2017 at: https://www.gov.uk/government/uploads/system/uploads/ attachment_data/file/609030/MHRA_GMP_Inspection_Deficiency_Data_Trend_2016.pdf
[9] EudraGMDP Non-compliance Report observed by the European Inspectors from February 2015 to May 2017, access on Jul 1, 2017 at: http://eudragmdp.ema.europa.eu/ inspections/gmpc/searchGMPNonCompliance.do
[10] Smith, “Rotational Cleaning-Is It Necessary?” Cleanroom Technology (January 2014)
[11] Martinez, JE “The Rotation of Disinfectant Principle True or False?” Pharmaceutical Technology (2009)
[12] Sutton, S. “Disinfectant Rotation in a Cleaning= Disinfection Program for Clean Rooms and Controlled Environments” CRC Press Taylor Francis (2007) edited by Gurusamy Manivannan
[13] Sutton, S. “Disinfectant Rotation a Microbiologists View” Cleanrooms, 2005, access on May 25, 2017 at: https:// cemag.us/article/2005/07/disinfectant-rotation- microbiologists-view
[14] PDA Technical Report Number 70: Fundamentals of Cleaningand Disinfection Programsfor Aseptic Manufacturing Facilities (2015)
[15] European Commission, Good Manufacturing Practice Medicinal Products for Human and Veterinary Use – Annex 1, Manufacture of Sterile Medicinal Products
[16] United States Pharmacopeia 38 (USP) <1072> – Disinfectantsand The United States Pharmacopeia Convention/National Formulary, Rockville, MD.
[17] Sartain E., Disinfectants rotation, access on Jul 1, 2017 at: https://www.cemag.us/article/2005/03/disinfectant- rotation
[18] FDA advises drug manufacturers that Burkholderia cepacia complex poses a contamination risk in non-sterile, water-based drug products, access on May 25, 2017 at: https://www.fda.gov/Drugs/DrugSafety/ucm559508. htm?source=govdelivery&utm_medium=email&utm_ source=govdelivery
[19] United States Department of Health and Human Services Food and Drug Administration, Guidance for Industry Sterile Drug Products Produced by Aseptic Processing — Current Good Manufacturing Practice, 2004.
[20] Regulatory Science of Pharmaceuticals and Medical Devices from Ministry of Health, Labour and Welfare of Japan, Guidance on the Manufacture of Sterile Pharmaceutical Products by Aseptic Processing, 2006.
[21] Rogers M. and Polarine J., Rinsing Strategy, access on August 25, 2017 at:http://www.ivtnetwcom/article/jim- polarine-and-marc-rogers-rinsing-strategy
[22] Sandle , aguidetocleaninganddisinfectingcleanrooms, 3rd edition, the CDC Handbook, 2012, p.173-180.

